Image made using the COMSOL Multiphysics® software and is provided courtesy of COMSOL.
A plate reactor where chemical reactions occur throughout and reacting chemicals are introduced at two points in the reactor.
Optimizing chemical reactors, filtration equipment, mixers, and other processes is made easy with the Chemical Reaction Engineering Module. It contains the tools for you to simulate material transport and heat transfer together with arbitrary chemical kinetics in all types of environments – gases, liquids, porous media, on surfaces, and within solid phases – or combinations of all of these. This makes it perfect for all facets of the chemical and process industries, and even within environmental engineering where the “process unit” or “chemical reactor” is the environment surrounding you.
The Chemical Reaction Engineering Module contains intuitive user interfaces for you to define material transport in dilute and concentrated solutions or mixtures through convection, diffusion, and ionic migration of an arbitrary number of chemical species. These are easily connected to definitions of reversible, irreversible, and equilibrium reaction kinetics that can be described by the Arrhenius equation, or any arbitrary rate law, where the effects of concentration and temperature on the kinetics can be included. The interface for defining chemical reactions is straightforward as chemical formulas and equations are entered essentially as you would write them on paper. COMSOL sets up the appropriate reaction expressions using the mass action law, which you can alter or override as you desire. The stoichiometry in your reaction formulas is used to automatically define mass and energy balances, either for homogeneous or heterogeneous reactor conditions; in bulk or on surfaces.
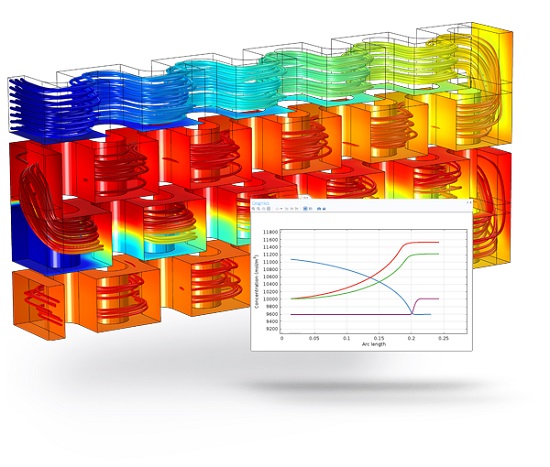
Chemical vapor deposition (CVD) allows a thin film to be grown on a substrate through molecules and molecular fragments adsorbing and reacting on a surface. This example illustrates the modeling of such a CVD reactor where triethyl-gallium first decomposes, and the reaction products along with arsine (AsH3) adsorb and react on a substrate to form GaAs layers.
The CVD system is modeled using momentum, energy, and mass balances including a detailed description of the gas phase and adsorption kinetics. A reduced reaction scheme is compared to the full scheme in the Reaction Engineering interface.
The model highlights the usability of the Reaction Engineering and Chemistry interfaces together with the Reversible Reaction Group feature for simulation of reaction/transport systems in 0 D and space-dependent reactors. In the Reaction Engineering interface you can easily study the transient behavior of different sets of reactions in a perfectly mixed system. The Chemistry interface collects reaction kinetics and calculates transport and thermal parameters, which can seamlessly be coupled with other interfaces.
In this application, you also utilize the Reversible Reaction Group feature for CHEMKIN import and organization of the complex system of bulk and surface reactions that are involved in the CVD process. The space-dependent model accounts for mass transport, heat transfer and fluid flow in the CVD reactor using the Transport of Diluted Species, Heat Transfer in Fluids, and Laminar Flow interfaces.
One of the most common reactors in the chemical industry, for use in heterogeneous catalytic processes, is the packed bed reactor. This type of reactor is used both in synthesis as well as in effluent treatment and catalytic combustion.
This model is set up to calculate the concentration distribution in the reactor gas that flows around the pellets, but it also uses an extra dimension that models the concentration distribution inside each porous catalytic pellet. The extra dimension is built in the feature Reactive Pellet Bed, which was introduced in COMSOL 5.0.
The reactor consists of a cylindrical shell filled with catalyst particles. These particles can be contained within a supporting structure, like tubes or channels, or they can be packed in one single compartment in the reactor.
This model presents an approach to studying coupled macro- and micro-mass balances in packed beds and other heterogeneous reactors. The model calculates the concentration profiles of reacting species both in the 3D packed bed reactor as well as in each individual catalyst pellet.
In this tutorial, the heat and mass transport equations are coupled to laminar flow in order to model exothermic reactions in a parallel plate reactor. It exemplifies how you can use COMSOL Multiphysics to systematically set up and solve increasingly sophisticated models using predefined physics interfaces.
The model simulates non-premixed turbulent combustion of syngas (synthesis gas) in a simple round-jet burner.
Syngas is a gas mixture, primarily composed of hydrogen, carbon monoxide and carbon dioxide. The name syngas relates to its use in creating synthetic natural gas. In the model, syngas is fed from a pipe into an open region with a slow co-flow of air. Upon exiting the pipe, the syngas mixes and combusts with the surrounding air in a non-premixed manner. The resulting turbulent flame is attached to the burner head.
The model is solved by combining the Reacting Flow and the Heat Transfer in Fluids interfaces. The turbulent flow in the jet is modeled using the k-ε turbulence model, and the turbulent reactions are modeled using the eddy dissipation model. The resulting velocity, temperature and species mass fractions in the reacting jet are compared to experimental values.
Carbon deposition on the surface of solid catalysts is commonly observed in hydrocarbon processing. A known problem is that carbon deposits can impede the activity of catalysts as well as block the flow of gas through a catalyst bed.
This example investigates the thermal decomposition of methane into hydrogen and solid carbon, over a catalyst. The influence on the catalyst activity is first studied with the Reaction Engineering interface in an ideal reactor model. This is extended to a space and time dependent model where the porosity decay due to carbon deposition is accounted for.
High-performance liquid chromatography (HPLC) is a common method of separating, identifying, and quantifying each chemical compound in a mixture. HPLC is found in the pharmaceutical, biotech, and food industries.
The Liquid Chromatography app simulates the separation of two species within a generic liquid chromatography column.
Simulations can help pinpoint design elements of chromatography columns and component separation details. This includes the length and porosity of the column and the specific conditions of the two components, including their Langmuir isotherm. All of this can be changed within the app.
This suite of examples illustrate the modeling of selective NO reduction, that occurs as flue gases pass through the channels of a monolithic reactor in the exhaust system of a motored vehicle. The simulations are aimed at finding the optimal dosing of NH3, the reactant that serves as reducing agent in the process.
Three different analyses are performed:
Kinetic analysis: The example takes a closer look at the selective reduction in a single channel of the monolithic reactor. The reaction kinetics is analyzed with the Reaction Engineering interface using the Plug flow reactor type.
Detailed process modeling: The example utilizes the optimal dosing of NH3as found in the above example as a starting point. This model is set up in 3D and can thereby reveal the full space-dependency of the problem with even better tuning of the dosing level.
Thermal stress modeling: The model studies thermal gradients in the monolith unit caused by heat released from the chemical reactions. The Structural Mechanics Module together with the 3d model above is used for this investigation.
This application presents an example of pressure driven flow and electrophoresis in a 3D micro channel system. Researchers often use a device similar to the one in this model as an electrokinetic sample injector in biochips to obtain well-defined sample volumes of dissociated acids and salts and to transport these volumes.
Focusing is obtained through pressure driven flow of the sample and buffer solution, which confines the sample in the focusing channel. When steady state has been obtained, the pressure driven flow is turned off and an electric field is applied along the channels.
The electric field drives the dissociated sample ions that are found in the focusing zone at right angles to the focusing channel, through the injection channel. This is solved in the time-domain.
This model requires the Chemical Reaction Engineering Module and either the CFD Module or the Microfluidics Module.
This application treats the flow field and species distribution in an experimental reactor for studies of heterogeneous catalysis. The model exemplifies the coupling of free and porous media flow in fixed bed reactors.
The reactor consists of a tubular structure with an injection tube that has its main axis perpendicular to the axis of the reactor. The incoming species in the main and injection tubes reacts in a fixed porous catalyst bed. The model couples the free fluid and porous media flow through the Navier-Stokes equations and Brinkman’s extension of Darcy’s law. The mass transport for the three species is modeled with the convection-diffusion equation.
Tubular reactors are often used in continuous large-scale production, for example in the petroleum industry. One key design parameter is the conversion, or the amount of reactant that reacts to form the desired product. In order to achieve high conversion, process engineers optimize the reactor design: its length, width and heating system. An accurate reactor model is a very useful tool, both at the design stage and in tuning an existing reactor.
This example deals with a gas-phase dissociation process, where species A reacts to form B. The model illustrates the use of several attractive features in the Chemical Reaction Engineering Module: